Successful Alopecia Areata Drug Trials!
The National Alopecia Areata Foundation estimated that 6.8 million Americans are affected by this auto-immune skin condition. With the large number of citizens affected, it is great news that clinical trials are showing success. HPIHair stays on top of these trials to keep our clients informed.
Phase 3 clinical trials using a common arthritis drug are showing that one in three patients with a severe skin disease were able to regrow hair after being treated with this drug.
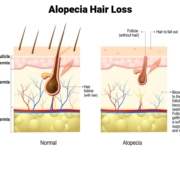
From Yale News: “The study is based on Phase 3 clinical trials using baricitinib, a Janus kinase (JAK) inhibitor, to treat alopecia areata, an often disfiguring skin disease characterized by rapid loss of scalp hair, and sometimes eyebrows and eyelashes.
Phase 3 clinical trials are the final testing hurdle before a new treatment can be considered for U.S. Food and Drug Administration (FDA) approval.
‘This is so exciting, because the data clearly show how effective baricitinib is,” said Dr. Brett King, an associate professor of dermatology at the Yale School of Medicine and lead author of the new study, published March 26 in the New England Journal of Medicine. “These large, controlled trials tell us that we can alleviate some of the suffering from this awful disease.'” (Read More Here)
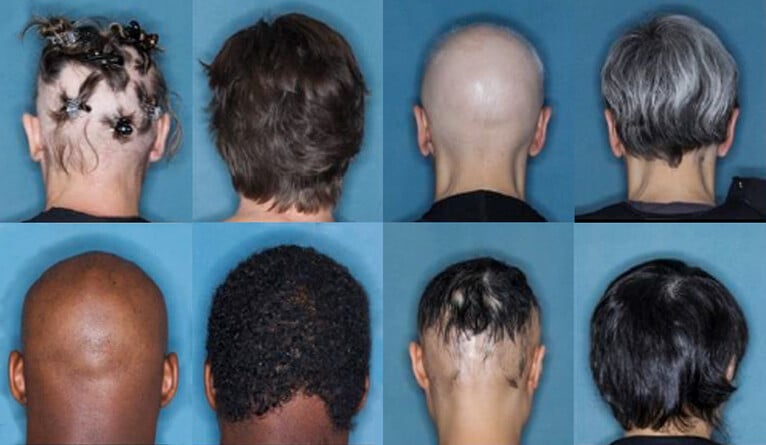
Photo Courtesy of Yale News: Before and after images for participants who received 36 weeks of treatment for alopecia areata with baricitinib.